The FDA has approved Anzupgo cream (delgocitinib) from Danish company LEO Pharma. It is the first treatment for chronic hand eczema in adults.
Pfizer is investing up to $15 billion to revamp its portfolio of obesity drugs after failing to complete trials for a drug for obesity. Under pressure from falling sales and criticism from major investors, the company has set a target of cutting costs by $7.2 billion by 2027.
The FDA has registered the first implantable gene therapy, Encelto. It slows the degeneration of the macula and prevents vision loss in type 2 macular telangiectasia.
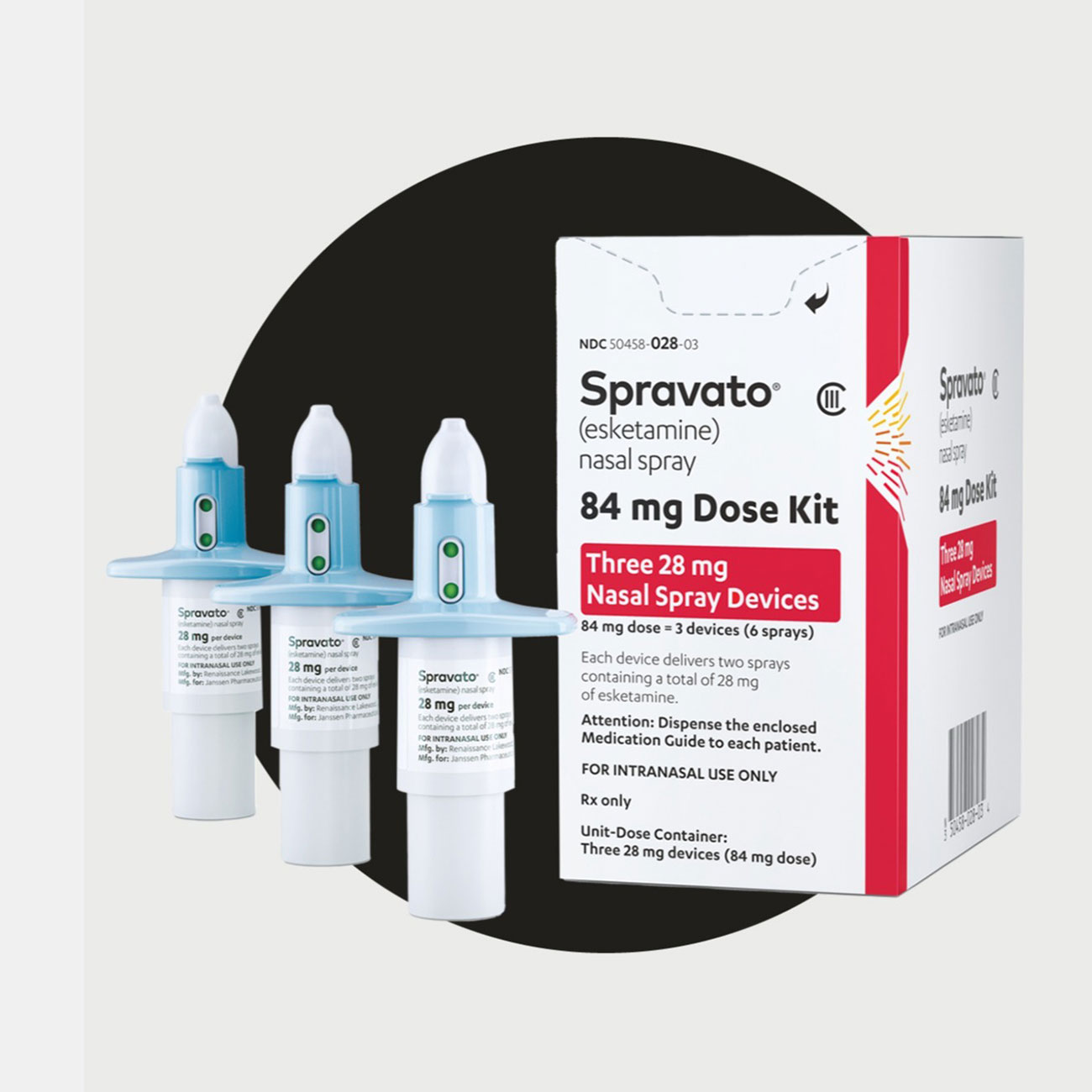
SPRAVATO® (esketamine) approved in the U.S. as the first and only monotherapy for adults with treatment-resistant depression
Following U.S. FDA Priority Review, approval is based on data demonstrating SPRAVATO® alone met its primary endpoint at 4 weeks and led to rapid and superior improvement in depressive symptoms compared to placebo as early as 24 hours.
Johnson & Johnson (NYSE:JNJ) December 16, 2024 announced the U.S. Food and Drug Administration (FDA) has issued a Complete Response Letter (CRL) for the Biologics License Application (BLA) for a fixed combination of amivantamab and recombinant human hyaluronidase for subcutaneous administration (SC amivantamab) in patients with non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutations.